|
3/17/2023 0 Comments Iso 14971 2007 free pdf  
Relevant information: reference to standards and other publications such as compatibility standards, regulatory authority guidance documents, and clinical literature. analogylsimilarity, demonstration, simulation, peer- review, testing or certification. 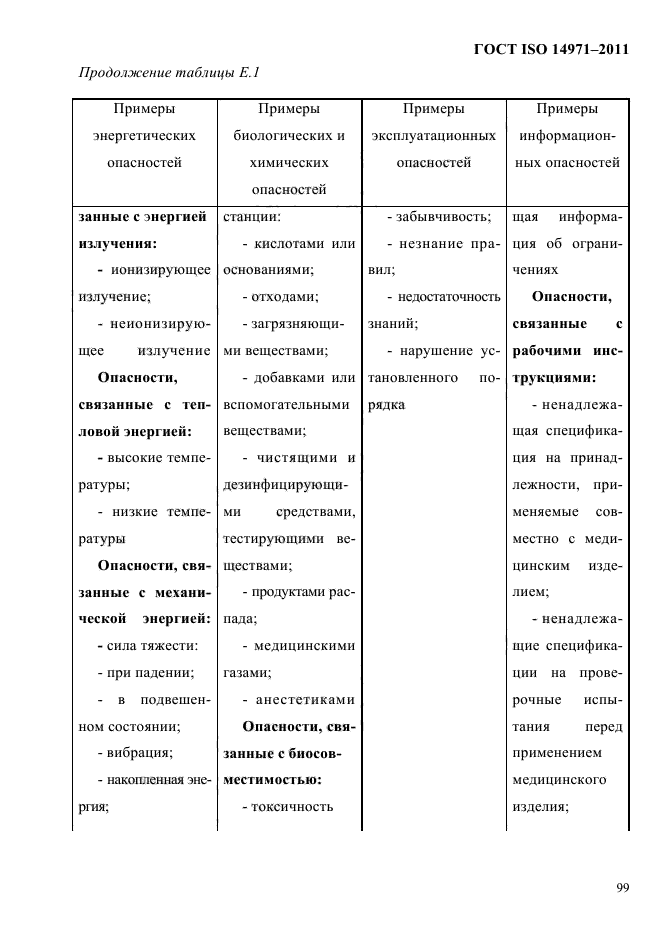
NOTE 2 VALIDATION methods include: inspection, analysis. NOTE I Constraints include: technical feasibility, cost, time, availability of VALIDATION enablers or qualified personnel, contractual constraints, criticality of the mission, etc. In the VALIDATION plan, the MANUFACTURER shall:Ī) identify the VALIDATION scope and the corresponding VALIDATION activities ī) identify the constraints that potentially limit the feasibility of VALIDATION activities Ĭ) select appropriate VALIDATION methods, input information, and associated acceptance criteria for successful VALIDATION ĭ) identify the enabling systems or services such as operating environment(s), including hardware and software platforms, needed to support VALIDATION Į) specify the required qualification of the VALIDATION personnel where training is required, this shall be completed before starting the VALIDATION į) define the appropriate level of independence of the VALIDATION team from the design team. The MANUFACTURER shall establish a VALIDATION plan addressing all HEALTH SOFTWARE PRODUCT use requirements established in 4.2. In this case, the MANUFACTURER shall take account of the RESIDUAL RISKS and implement RISK CONTROLS around those found to be unacceptable. It is recognized that the MANUFACTURER might not be able to follow all the process steps identified in ISO 14971:2007 for each constituent component of the HEALTH SOFTWARE, such as proprietary components, subsystems of non-healthcare origin, and legacy software. The requirements in 4.2, 4.3, Clause 5, Clause 6, Clause 7, Clause 8 and Clause 9 of IEC 62304:2006 and IEC 62304/AMD1:2015 shall apply to the HEALTH SOFTWARE in addition to the other requirements of this document. The system requirements for the HEALTH SOFTWARE PRODUCT established in 4.5 shall be used as primary design input for the life cycle process of the HEALTH SOFTWARE PRODUCT. IEC 82304-1 pdf free download.Health software- Part 1: General requirements for product safety.ĥ * HEALTH SOFTWARE - Software life cycle processes 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
